The word corrosion derives from Latin ‘corrodere’ which translates to ‘gnaw to pieces’. For marine applications, mild steel remains the number one metal for constructional purposes, its main drawback being that it corrodes easily in seawater. Preventing corrosion requires elimination or suppression using cathodic protection and/or coatings. Repairing coatings offshore can be up to 100 times the cost of the initial coating so it pays to get it right the first time.
What is corrosion?
Corrosion is a natural process of materials, usually metals, moving towards their lowest possible energy state, resulting in a spontaneous reaction between the material and its environment which results in the degradation of that material. The word derives from Latin ‘corrodere’ which translates to ‘gnaw to pieces’.
For marine applications, mild steel remains the number one metal for constructional purposes by virtue of its relatively low cost, mechanical strength and ease of fabrication. Its main drawback is that it corrodes easily in seawater and unless adequately protected, rapidly loses strength which may result in structural failure. The diagram below demonstrates the cycle of corrosion. From the mining of iron oxide, producing steel, to corrosion.
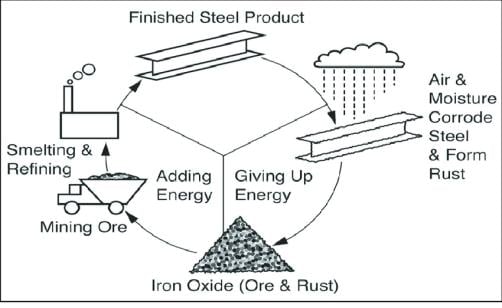
Image: ResearchGate
Repairing coatings offshore can be up to 100 times the cost of the initial coating, and NACE International estimates that the total cost of marine corrosion worldwide is between $50-80 billion every year. Source: Maritime Industry. 2018. Maritime Industry. [ONLINE] Available at: https://www.nace.org/Corrosion-Central/Industries/Maritime-Industry/.
With proper planning, ship-owners can ensure that their ships are operating at maximum performance and maintain cost efficiency while preserving the condition of their assets. If poor surface preparation is the cause of deterioration, then the only solution is to remove the paint and start again. It pays to get it right the first time.
Two types of corrosion particularly relevant for the marine industry are pitting corrosion and bacterial corrosion.
How to prevent corrosion
Preventing corrosion requires elimination or suppression using two principal methods, cathodic protection and coatings. Generally, cathodic protection systems are used in conjunction with coating systems.
Cathodic protection
The objective with cathodic protection is to suppress the electrochemical reaction taking place. Under normal corrosive conditions, current flow from the anode results in a loss of metal at the anodic site which results in the protection of the metal at the cathodic site.
Protection can be provided by making the structure you wish to protect cathodic, using two methods:
1. Sacrificial anodes:
When two dissimilar metals are immersed in seawater, the metal with the lowest electrical potential will suffer the greatest corrosion. For example, the corrosion rate of mild steel can be controlled by connecting it to zinc as it will then become the anode and corrode. In this example, the zinc anode is referred to as a sacrificial anode because it is slowly consumed (corrodes) during the protection process.
Another use of zinc as a sacrificial anode is when steel is coated with the zinc; either in the form of galvanization or metalisation or in a paint which contains high levels of active zinc.
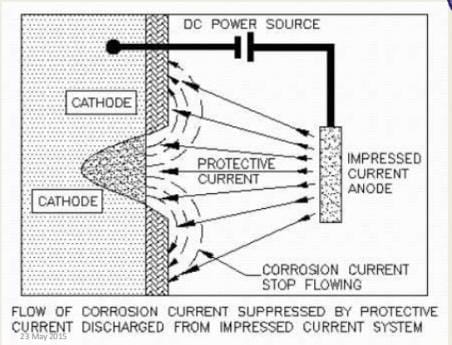
2. Impressed current systems:
A vessel hull can be made cathodic by using a direct current source. An impressed current is applied in the opposite direction to cancel out the corrosion current and convert the corroding metal from anode to cathode. In this example, the negative terminal of DC is connected to a pipeline to be protected. The anode is kept in to increase the electrical contact with its surrounding environment.
Coatings
The effectiveness of coatings preventing corrosion depends upon many factors, for example, coating type, end-use of coating, and vessel operating environment.
Low permeability and good ‘wet adhesion’ i.e. adhesion under immersion, are widely believed to be the single most important aspects of corrosion control by coatings.
- Adhesion
For maximum coatings adhesion, steel surfaces must be clean, dry and free of oil, rust, salts and other contaminants before painting.
- Permeability
Highly cross-linked, chemically curing systems are likely to have relatively low permeability characteristics and film thickness can affect it. In general, thicker films delay the passage of oxygen and water to the steel surface. High film thickness (>400 um dft) can therefore offer a high degree of corrosion protection which would best be achieved in multi-coat systems rather than in a single coat.
- Damaged coatings
Coatings in service may be subjected to mechanical damage. Maintaining corrosion protection can therefore best be achieved by using coatings which offer both abrasion resistance and corrosion protection. It is recommended that coatings have good ‘undercutting’ resistance i.e. resistance to under-film corrosion creep at damaged areas.
Other mechanisms used to prevent corrosion in non-immersed, above water areas include:
- The use of anticorrosive pigments such as zinc phosphate. This pigment is slightly soluble and can form a corrosion inhibiting molecular layer at the steel surface.
- The use of metallic zinc as a sacrificial pigment, in essence the development of an “in situ” cathodic protection system.
Our success with corrosion prevention:
Many AkzoNobel customers have had success preventing corrosion with Intershield 300. Read more:
- ‘Samco Raven’ soars with Intershield 300 after 15 years in service.
- Stolt Tanker benefits from Intershield with zero repairs need in the first 20 years of service.
- The 20,000th application of Intershield 300
- Learn about our Intertrac OBM digital tool to help you optimise OBM purchasing and improve and maintain your vessels
Contact us
Contact us if you would like more information about how to prevent corrosion, or contact your sales representative.
Contact us

We are here to help
Speak with one of our experts to discuss how we can get the best results for your area of application.